A few months ago I wrote about a new lithium mine at Thacker Pass which is in northern Nevada near the Oregon border. The geologically important area is called the McDermitt Caldera which is an ancient, collapsed volcano, much like Crater Lake (just not filled with water!). It turns out that some folks took another look at the area and decided it was richer in lithium than they thought. In fact, it might be the largest lithium deposit in the world.
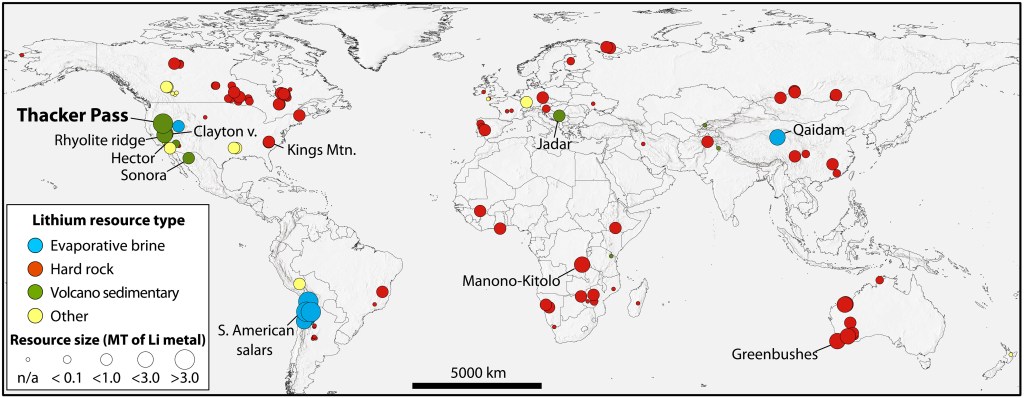
Lithium is often extracted from subsurface brines. These fluids are pumped up out of the ground like oil. Unlike oil they are spread out into shallow ponds and the water allowed to evaporate (much like obtaining sea salt) leaving the minerals behind. The salts are processed to separate out the desired minerals. Lithium is usually marketed as lithium carbonate. Here’s a look at lithium distribution in the world:
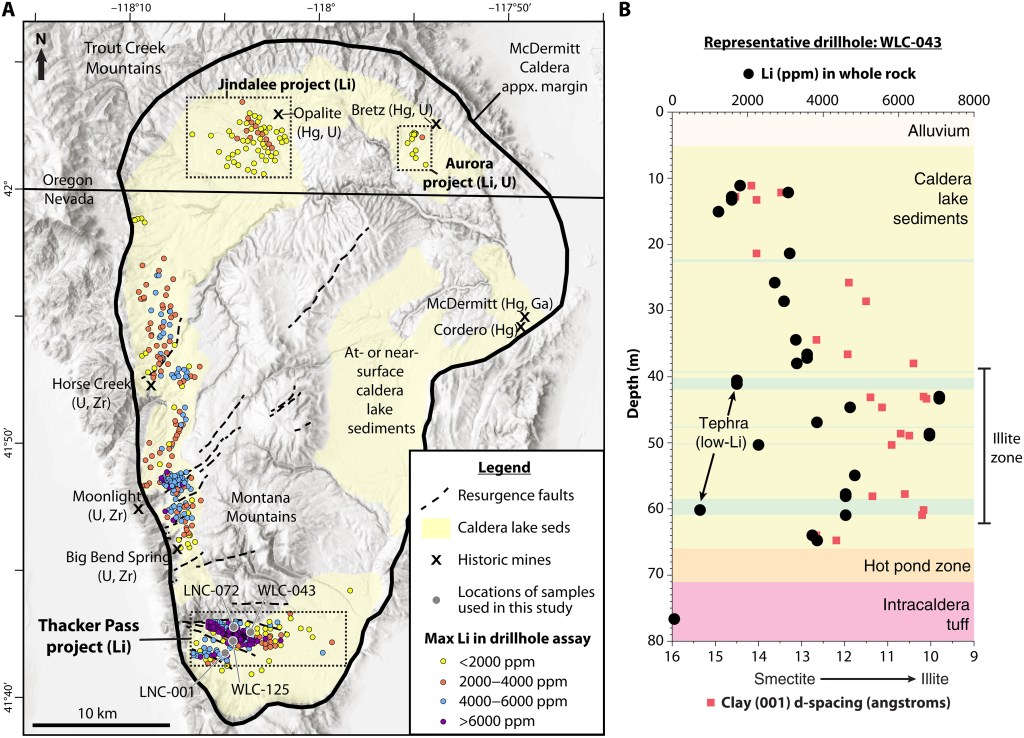
This lithium deposit in the McDermitt Caldera is composed of clays on or near the surface. Specifically they are “clay-rich lacustrine sediments” (lacustra is Latin for lake) that were laid down in Miocene times (5-23 million years ago). Obviously the climate was much different, today it is a vast desert, then it was probably sub-tropical. Illite and smectite are the primary ores, both are sheet silicates. The mining will be done by open pit methods. You can see on the graphic below that the clay sediments are within tens of meters of the surface:
There is enough lithium in the world to supply automakers with this crucial EV battery material. But there isn’t much in the way of domestic supply, hence the excitement about this deposit in Nevada. It is potentially large enough so that American companies would not have to import any lithium.
During WWII there was not enough domestic uranium to supply the Manhattan Project. About two-thirds of the stuff came from a mine in the Belgian Congo. Thus there is always a strategic interest in local sources of critical minerals.
We are committed to electrifying our vehicle fleets. In the long run this will be a good thing. But EVs have created new demand for things like copper, zinc, and lithium and we’ll have to find new sources (mines!) for them. And there should be incentive to re-visit previously worked sites and to recover new minerals by recycling. And I think it is desirable to have domestic mines because we have, at least nominally, the rule of law here and thus environmental regulations actually have a chance of being enforced. If we are going to make big messes in the ground, let’s at least do it where we can keep an eye on it and clean up after it.
EVs are still vehicles. They still need roads. They still need steel, glass, aluminum, and plastic. And they still need parking lots! There will still be traffic snarls, and congestion, and long, soul-crushing commutes. A cleaner car or truck is better than a dirtier one, but it doesn’t address the real problems. Do we still want a future where cities are increasingly uninhabitable for pedestrians? Do we still want a future where you have to drive everywhere? Do we still want a future where other transit options continue to fade away? We are a car-centric culture, and that car-centrism has shaped the way we live, work, and play. Is it possible to imagine other ways to live, especially ways that don’t require each of us to own and maintain a 5000-pound metal behemoth?
Before we get too excited about future vehicles, maybe we should spend more time thinking about the kind of future we want to live in.
