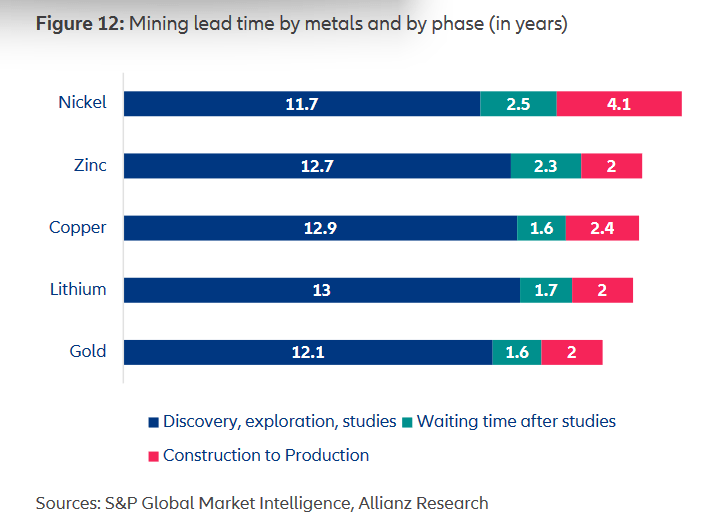
Color TV broadcasts started in this country in the 1950s but really didn’t take off until the mid-60s. In 1972 half of the television sets in the US were color. I remember our family getting a color TV (and cable!) right about then. Very few all black-and-white TV stations remained by the end of that decade.
One of the barriers to color reception was red. Color is displayed on screens by means of phosphors. These are materials that emit light when exposed to radiant energy. In a TV, an electron beam activates the luminescent substance. Different chemicals give off different colors.
In the case of red, the key breakthrough was the use of the lanthanoid or rare-earth element Europium. Yttrium orthovanadate (YVO4) is “doped” with a small amount of europium oxide (Eu2O3) to produce a bright red glow. TV sets contain about a half a gram to a gram of Eu2O3.
The European Union takes advantage of europium oxide’s luminescent property by embedding it in fibers in their banknotes. This is an anti-counterfeiting measure. You can see the red stripe that emerges under UV light in the 100-euro bill:
Europium has no known biological role and is not toxic. Europium is one of the rarest of the lanthanoids. In the Mountain Pass Mine in California, our only domestic source of REEs (rare earth elements), europium is a tiny fraction (0.2%) of the ore.