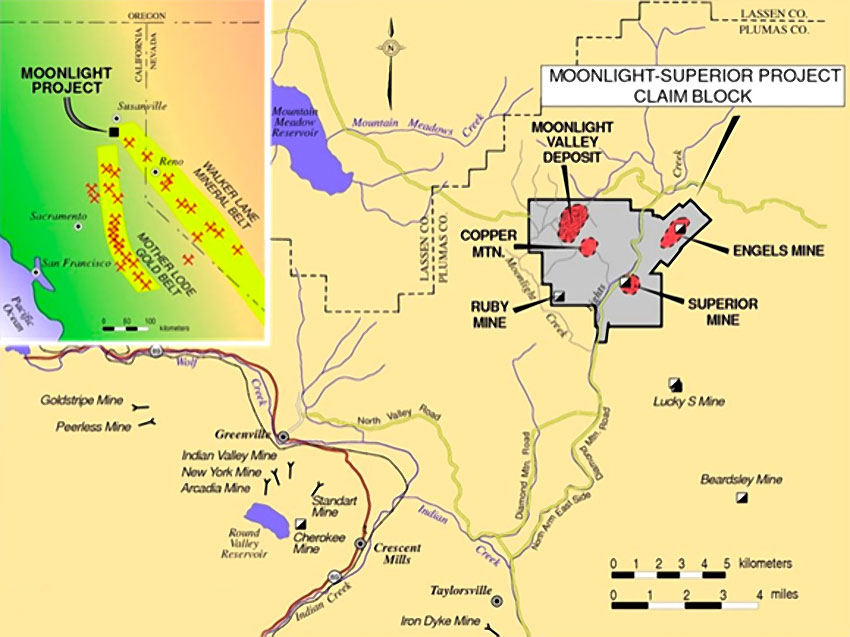
There wouldn’t be much to Las Vegas without neon. We are all familiar with the orange-red glow of neon from the many signs we’ve seen our whole lives. “OPEN” and “VACANCY” are probably the most common but of course every bar and lounge in the country has neon-sign beer adverts festooning the walls and beckoning customers from the windows.
Gas discharge tubes work by filling a vacuum space (like a glass tube) with a substance (like neon gas) at very low pressure. Electric current is passed through the gas in the tube and you get ionization. Some of the gas atoms lose an electron and these electrons migrate to the anode (one end of the tube). The gas ions formed migrate in the electric field towards the cathode (the other end of the tube). On the way they collide with neutral atoms and re-gain an electron. This lowers their energy state and a photon of light is emitted. Most of the photons are in the orange-red spectrum and that’s what we see.
Neon is a so-called “noble” gas. The far right column of the periodic table (group 18) contains the family of inert gases: helium, neon, argon, krypton, xenon, and radon. All are interesting chemically because their outermost electron shells are filled and thus they are not very reactive. It takes a lot of energy or unusual circumstances for these atoms to form compounds. They do ionize however, and when all the atoms in a gas become ions the material is known as plasma. This is often thought of as the fourth state of matter after solids, liquids, and gases. Much of our universe—stars, for example—are plasmas. Lightning is a plasma. Neon and mercury vapor mixtures were used in the tiny cells of the first plasma television sets. Similar gas mixtures form the plasma in fluorescent lighting.
The next time you look at a neon sign beckoning you to drink, gamble, or both, think about plasma. After all, we are stardust. The atoms that make us up were born in stars a long time ago. And to stardust we will eventually return. First the earth will recycle the atoms in our bodies. Some long time later galaxies will collide and recycle whole solar systems. Perhaps on some distant planet in the far future a primitive being will build a device that excites the atoms that once were part of you and they will glow with the colors of the universe.