Take a look at the periodic table of the elements:
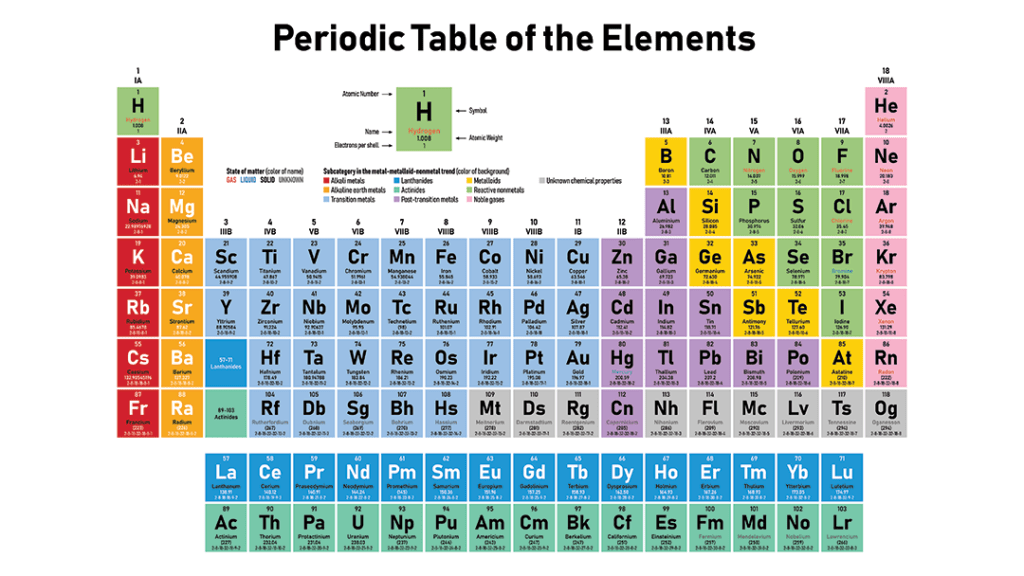
You will find Yttrium (symbol “Y”) just below Scandium (#21, Sc) in column three. Beneath that you see the box for the lanthanides, elements 57 through 71. You have to imagine that row of blue (La to Lu) squeezed into the space between Barium (Ba, #56) and Hafnium (Hf, #72). I’ve even seen it represented in 3-D with the blue row of lanthanides popping out of the page toward the reader.
Columns on the periodic table are called “groups.” Elements in the same group have similar properties. This is due to their electron arrangement. All the elements in group one, for example, have one electron in their outer shell and this makes them highly reactive. They readily form positive ions. All the way to the right, in group eighteen, the elements have filled outermost electron shells and are thus inert. They don’t readily ionize.
You’ll see that the element yttrium is in the same column as the element lanthanum, #57, the “head” of the group called lanthanides. We talked about this group in a previous post. These materials, for historical reasons, are called “rare earths.” Yttrium and lanthanum have the same outer shell electron configuration and thus have similar properties. Although it is not technically a “rare earth” like lanthanum and the rest of that row (57-71) it is close enough to be classified with them.
Yttrium is more abundant than silver, being found in the earth’s crust at 31 parts per million. Silver checks in at significantly less than 0.1 ppm. Yttrium, like all the lanthanides, is never found in nature as a pure metal. It is mined from the same sources as the rest of the rare earths.
Yttrium is used in video displays to make red colors. It is used to make synthetic garnets and as an alloying agent for magnesium and aluminum. It is also used in lasers. Yttrium, like erbium, terbium, and ytterbium, is named for the village of Ytterby in Sweden.
The United States consumes about 700 metric tons of yttrium annually.
Thanks for the update, Mr. Science!
LikeLike
Anytime.
LikeLike
[…] of Resarö which is part of the Stockholm archipelago. It is immortalized on the periodic table. Yttrium (#39), Terbium (#65), Erbium (#68), and Ytterbium (#70) are all named for Ytterby. A centuries-old local […]
LikeLike